CIIC Spring Conference Page
Improving Biologic Infusion Preparation Through Controlled Air Equilibrium
Early Clinical and Operational Observations Using Aeristasis™ Technology with I2F®
Presenting Authors: Damea Alexander; Maeve O’Connor, MD, FACAAI, FAAAAI, FACP
Watch: What we mean by Controlled Air Equilibrium
Clinical comparison: conventional preparation vs I2F
Observe the difference:
• Left: Conventional vented spike workflow
• Right: I2F with Aeristasis™ controlled air equilibrium
• Note foaming, pressure behavior, and medication retention
Want to review this later?
We’ll open your email app so you can send this page to yourself.
Welcome to CIIC 2026 Spring Conference
Thank you for connecting with us at the Consortium of Independent Immunology Clinics (CIIC) meeting. This page is dedicated to clinicians, operators, and innovation leaders who want to learn more about Aeristasis™ Technology with I2F and its potential impact on biologic infusion preparation.
Our work focuses on improving infusion preparation through controlled air equilibrium — supporting protein stability, reducing foaming, and improving workflow efficiency during multi‑vial biologic infusions.
Clinical Context
During biologic infusion preparation, air flow into vials during withdrawal can contribute to:
Foaming and protein stress
Preparation inefficiencies
Recurrent bubbles during handling of plasma- and protein-derived therapies
Foaming and agitation of protein-based therapeutics are recognized contributors to protein aggregation, which may impact drug stability and performance. These effects can recur across infusions, creating cumulative medication loss and operational burden.
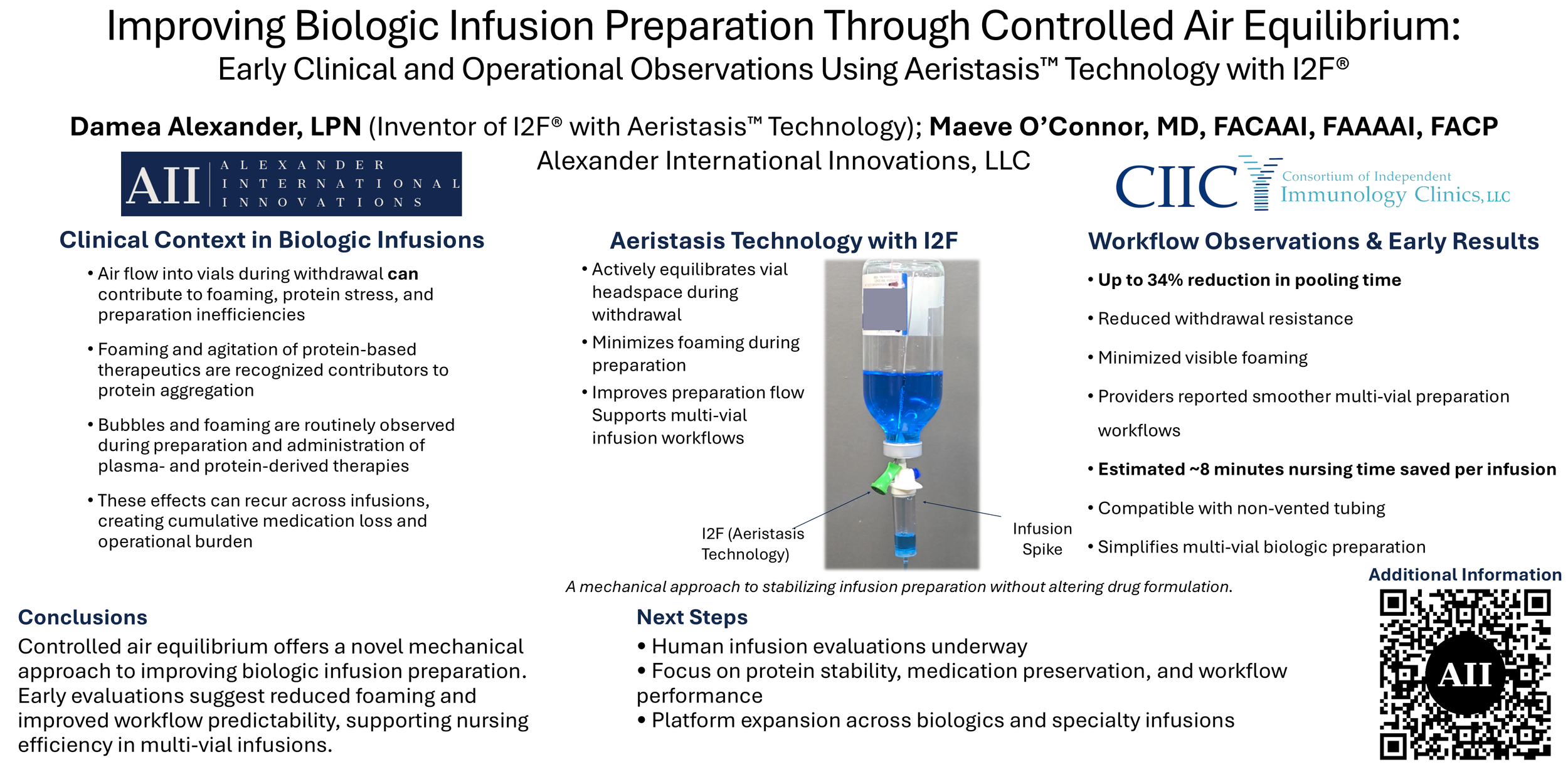
Aeristasis™ Technology with I2F®
I2F — powered by Aeristasis Technology
Controlled air equilibrium at the point of vial access for predictable medication preparation
Aeristasis Technology with I2F provides a mechanical approach to actively equilibrating vial headspace during medication withdrawal — without altering drug formulation.

Key Capabilities
Actively equilibrates vial headspace during withdrawal
Minimizes foaming during preparation
Improves preparation flow
Supports multi-vial infusion workflows
A mechanical approach to stabilizing infusion preparation without altering drug formulation.
I2F used alongside a standard infusion spike to equilibrate vial headspace during preparation.
Workflow Observations & Early Results
Early workflow simulations and clinical-use evaluations in veterinary infusion settings demonstrated:
Up to 34% reduction in pooling time
Reduced withdrawal resistance
Minimized visible foaming
Providers reported smoother multi-vial preparation workflows
Estimated ~8 minutes nursing time saved per infusion
Compatibility with non-vented tubing
Simplified multi-vial biologic preparation
Conclusions
Controlled air equilibrium offers a novel mechanical approach to improving biologic infusion preparation. Early evaluations suggest reduced foaming and improved workflow predictability, supporting nursing efficiency in multi-vial infusions.
Injectable Preparation: Reducing Spray Loss from Over‑Pressurization
While this poster focuses on biologic infusions, the same air‑equilibrium principles apply to injectable medications. During conventional vial access, pressure differentials can lead to visible spray or misting when needles are removed or doses are withdrawn — contributing to unintended medication loss, environmental contamination, and inconsistent dosing.
Aeristasis Technology with I2F supports controlled air equilibrium during vial access, helping to reduce pressure-driven spray events and improve medication handling consistency during injectable preparation.
This expanded application area represents an additional opportunity to:
Reduce high‑cost injectable medication loss
Improve dose accuracy
Support safer preparation environments
Example of pressure-driven spray during conventional vial access. Controlled air equilibrium helps reduce these events during preparation.
How to use I2F with a needle/syringe
Technical & Clinical White Papers
-
Explores how air bubble formation during medication preparation contributes to protein agitation, denaturation, and aggregation in biologics, and how I2F’s controlled air equilibrium may help reduce bubble-related stress at the point of vial access.
-
Examines infusion-related contributors to IVIG-associated aseptic meningitis, including pressure instability and microbubble introduction, and discusses how improved air management during preparation may support safer infusion practices.
-
Reviews potential mechanisms behind infusion-related hypersensitivity reactions in immunoglobulin therapy and evaluates how minimizing air introduction and pressure variability during vial access may improve medication tolerability.
-
This white paper explores how controlled air equilibrium during infusion preparation may support payer priorities by:
Helping preserve high-cost biologic medications by reducing foaming and handling-related losses
Improving preparation efficiency and nursing workflow during multi-vial infusions
Supporting more consistent medication handling practices across infusion sites
Enabling potential cost savings through improved operational efficiency and reduced medication loss
Aligning drug delivery practices with goals of cost containment, efficiency, and quality of care
-
Examines growing evidence of microplastic exposure from plastic-based medical delivery systems and introduces stainless steel vial access as a potential pathway to reduce particulate burden during injectable and infusion workflows.